What is C-CDA (Consolidated Clinical Document Architecture)?
C-CDA (Consolidated Clinical Document Architecture) is a healthcare data standard developed by HL7 (Health Level Seven International) for creating structured electronic clinical documents. C-CDA defines both the format and content of documents such as discharge summaries, progress notes, consultation reports, and care plans, ensuring that health information can be exchanged consistently between different electronic health record (EHR) systems.
C-CDA builds on earlier CDA standards by consolidating them into a single framework, making it the most widely adopted U.S. standard for clinical document exchange. It encodes information with standardized terminologies like SNOMED CT for clinical concepts, LOINC for lab and observation results, and UCUM for units of measure.
Although newer standards like FHIR (Fast Healthcare Interoperability Resources) are becoming more prominent, C-CDA remains central to interoperability, especially in regulatory reporting, transitions of care, and health information exchange networks.
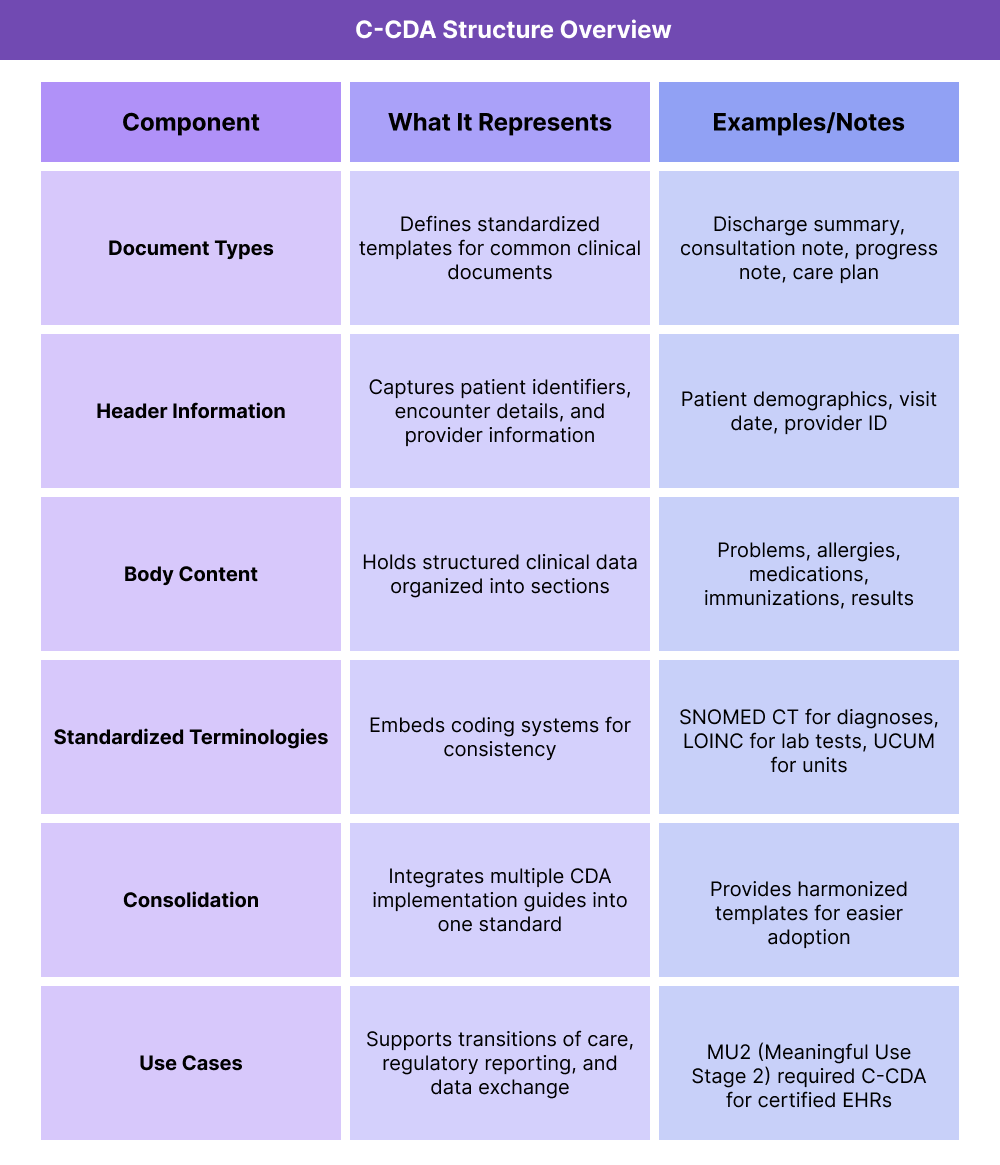
Key Components of C-CDA
C-CDA Document Types
C-CDA provides a standardized set of document templates that cover the most common clinical communication needs. These include discharge summaries, consultation notes, progress notes, and care plans. Each template follows a uniform structure, ensuring consistency in how information is captured and shared.
Header Information for C-CDAs
Every C-CDA document begins with a structured header that contains essential metadata. This includes patient identifiers (such as name, date of birth, and medical record number), encounter details (visit type, admission/discharge dates), and provider information (organization, clinician name, and credentials). The header ensures that documents can be matched to the correct patient and episode of care when exchanged across systems.
Body Content of a C-CDA
The body of a C-CDA document is divided into clinical sections, each with its own standardized format. Common sections include problems, allergies, medications, immunizations, procedures, and results. Because these sections are structured, receiving systems can parse and incorporate the data rather than just storing it as unstructured text.
Standardized Terminologies
C-CDA leverages recognized coding systems to ensure semantic accuracy across platforms. For example, SNOMED CT is used for problems and diagnoses, LOINC for lab tests and observations, and UCUM for units of measure. This linkage allows different systems to interpret the meaning of the data in the same way, even if they use different internal vocabularies.
Consolidation
Before C-CDA, multiple CDA implementation guides existed for different types of documents, creating complexity for implementers. C-CDA unified these guides into a single consolidated standard, harmonizing templates and reducing duplication. This made adoption more practical and consistent across vendors and providers.
C-CDA Use Cases
C-CDA is widely used in real-world healthcare workflows. It supports transitions of care, enabling one provider to send a standardized summary to another when a patient is referred or discharged. It is also used in regulatory programs such as Meaningful Use and Promoting Interoperability, which required certified EHRs to generate and consume C-CDA documents. Additionally, C-CDA underpins many health information exchange (HIE) networks that rely on document-based interoperability.
How C-CDA Is Used in Practice
Step 1 — Document Creation
When a clinical encounter occurs (such as a hospital discharge, outpatient consultation, or routine visit), the provider’s EHR generates a C-CDA document. This document includes both the header (patient and encounter details) and the body (clinical data such as problems, medications, allergies, and results).
Step 2 — Encoding with Standard Terminologies
Within the document, data elements are encoded with standardized vocabularies. For example, diagnoses are captured in SNOMED CT, labs in LOINC, and measurement units in UCUM. This ensures that receiving systems interpret the data consistently.
Step 3 — Document Transmission
The C-CDA file is packaged in XML format and exchanged between systems. This may happen via direct secure messaging, through a health information exchange (HIE), or as part of regulatory reporting workflows.
Step 4 — Integration into Receiving Systems
When the receiving EHR ingests the C-CDA, it parses the structured sections. Instead of treating the file as a flat PDF or scanned note, the system can directly incorporate the discrete data — such as updating a patient’s medication list or allergy record.
Step 5 — Use in Care Coordination
Providers use C-CDA documents to share patient summaries during transitions of care. For example, when a primary care physician refers a patient to a specialist, the specialist receives a structured summary that includes recent lab results, active problems, and treatment history.
Step 6 — Use in Compliance and Reporting
C-CDA also plays a role in regulatory programs. Under Meaningful Use (now Promoting Interoperability), certified EHRs were required to support C-CDA generation and exchange. Many quality reporting and public health initiatives also continue to accept C-CDA-based submissions.
Billing and Reimbursement for C-CDAs
Is C-CDA Used for Billing?
Not directly. C-CDA is not a billing code set like ICD-10 or CPT. Instead, it is a document standard that packages and transmits clinical information.
While claims themselves are typically transmitted using X12 EDI formats, C-CDA can include coded data (diagnoses, procedures, results) that supports downstream billing accuracy.
How C-CDA Influences Reimbursement
- Accurate coding context: Because C-CDA documents embed standard terminologies (SNOMED CT, LOINC, UCUM), they help ensure diagnoses and procedures are consistently represented, which supports accurate billing when mapped to ICD-10 or CPT.
- Value-based care alignment: C-CDA documents are used in quality reporting and regulatory programs (e.g., Promoting Interoperability), which tie into incentive payments or penalties.
- Care transitions: Structured patient summaries reduce information gaps that could otherwise lead to duplicative or denied services.
Limitations of C-CDA in Billing
- Not a billing standard: Claims processing does not rely on C-CDA — it must be supplemented with other formats (X12, UB-04, CMS-1500).
- Workflow burden: Generating and transmitting C-CDA documents requires EHR integration, which may be inconsistently implemented across vendors.
- Transition to FHIR: As FHIR adoption grows, C-CDA is increasingly seen as a legacy standard, especially for real-time data exchange, though it remains widely used in regulatory programs.
Future Outlook
- C-CDA will continue to serve as a compliance backbone for regulatory reporting and health information exchanges in the near term.
- Over time, FHIR APIs are expected to take on many of the same use cases, especially for payer-provider data exchange and patient access mandates.
Quality & Equity Implications of C-CDAs
Improving Data Quality
C-CDA enforces structured formats and standardized vocabularies, which improves the accuracy of patient records. By encoding problems, medications, and results in SNOMED CT, LOINC, and UCUM, the data remains consistent across different EHR systems. This reduces errors and makes patient information more reliable for clinical decision-making.
Supporting Care Coordination
C-CDA documents provide a comprehensive patient snapshot during transitions of care. A specialist receiving a referral, or a hospital discharging a patient, can share structured summaries instead of relying on faxed notes. This improves continuity, reduces duplication of services, and supports better patient outcomes.
Regulatory and Quality Reporting
C-CDA plays a key role in U.S. compliance programs. Under Meaningful Use (now Promoting Interoperability), certified EHRs had to generate and exchange C-CDA documents for core quality measures. Even as FHIR APIs take on more reporting roles, C-CDA remains a foundation for regulatory data submissions.
Equity Considerations
- Visibility into disparities: By embedding standardized codes for conditions, labs, and social factors, C-CDA helps capture data that can be aggregated for disparity monitoring.
- Enabling quality comparisons: Because documents are structured, organizations can compare outcomes across different populations, informing equity-driven interventions.
- Limitations: C-CDA itself does not mandate collection of social determinants of health (SDOH) data. Its ability to support equity depends on what information providers choose to document.
Challenges
- Legacy format: While still widely used, C-CDA lacks the flexibility of FHIR, limiting its ability to support modern analytics and patient-facing apps.
- Adoption variability: Not all providers generate complete or high-quality C-CDA documents, leading to gaps in data exchange.
Frequently Asked Questions about C-CDA
1. What is C-CDA in healthcare?
C-CDA (Consolidated Clinical Document Architecture) is a standard developed by HL7 for structuring and exchanging electronic clinical documents. It ensures patient information can be shared consistently between different EHR systems.
2. What types of documents are included in C-CDA?
C-CDA covers common healthcare documents such as discharge summaries, consultation notes, progress notes, care plans, and referral summaries.
3. Can you give an example of a C-CDA document?
Yes. A hospital discharge summary formatted in C-CDA includes a structured header with patient identifiers and encounter details, plus body sections like problems, medications, allergies, and lab results.
4. Is C-CDA used for billing?
Not directly. C-CDA is not a billing code set. However, it carries structured clinical data (diagnoses, procedures, labs) that can be mapped to ICD-10 or CPT codes to support accurate billing and quality reporting.
5. How is C-CDA different from FHIR?
C-CDA is a document-based standard that exchanges complete patient summaries. FHIR is a newer, API-based standard that exchanges modular data elements in real time. Both are HL7 standards, and they are often used together.
6. Is C-CDA required by CMS or federal regulations?
Yes. C-CDA was mandated under the Meaningful Use / Promoting Interoperability programs for certified EHRs. While FHIR is now being prioritized for patient access and payer data exchange, C-CDA remains a compliance requirement in many regulatory workflows.
