CPT 98985 Description, Billing Rules, & Use Cases
CPT 98985 is used to report Remote Therapeutic Monitoring (RTM) device supply for monitoring of the musculoskeletal system when patient-generated RTM data is captured or transmitted on 2 to 15 days within a 30-day period. RTM for musculoskeletal conditions is commonly used to track therapy adherence, therapy response, and digital therapeutic interventions, such as home exercise programs or post-operative rehabilitation activities performed outside the clinic.
CPT 98985 reimburses the device supply and data access/transmission layer of a musculoskeletal RTM program. This includes furnishing the RTM device or digital therapeutic tool, enabling data collection, and maintaining access to musculoskeletal therapy-related data for clinical oversight. The code is designed for short-duration monitoring when the patient does not meet the 16-day threshold required for the standard musculoskeletal RTM device supply code.
CPT 98985 does not represent provider treatment management time. Clinical review, patient communication, and therapy-related decision-making are reported separately using RTM treatment management codes when time and communication requirements are met.
What is CPT Code 98985?
CPT 98985 covers the monthly supply of an RTM device or digital therapeutic tool used to support musculoskeletal system monitoring. It is billed once per 30-day period when:
- A qualified RTM device or digital therapeutic tool is supplied to the patient
- The RTM system supports musculoskeletal therapy monitoring (e.g., exercise adherence, functional response, rehabilitation progress)
- RTM data is captured and available on 2–15 days within the billing cycle
CPT 98985 functions as the infrastructure code for musculoskeletal RTM. It captures the technical services that support therapy monitoring—such as device access, data transmission, and system connectivity—when the patient’s monitoring activity is clinically meaningful but does not reach a 16-day threshold.
This code does not include:
- RTM setup or onboarding services (if applicable under your workflow)
- RTM treatment management time (reported separately under CPT 98979 or CPT 98980)
CPT 98985 is billed instead of the full-duration musculoskeletal RTM device supply code for the same system and billing period and should not be reported concurrently with that code.
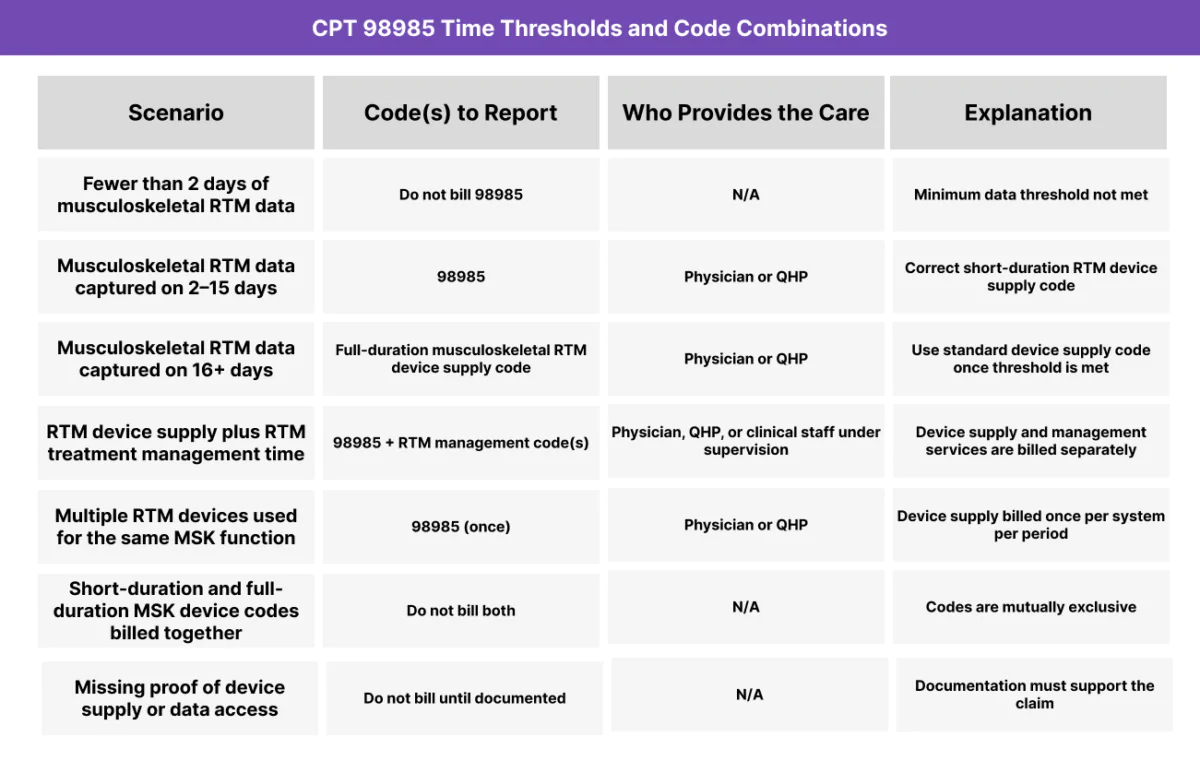
CPT 98985 Time Thresholds and Code Combinations
CPT 98985 may be billed once per 30-day period to report RTM device supply and data access/transmission for musculoskeletal system monitoring when RTM data is available on 2–15 days.
Important to Note:
CPT 98985 requires:
- RTM device supply tied to musculoskeletal system monitoring
- RTM data availability on 2–15 days within the 30-day period
- One billing instance per patient, per 30-day cycle, for musculoskeletal monitoring
- Documentation supporting device supply and data access/transmission
When to Use CPT 98985: Common Scenarios and Use Cases
CPT 98985 applies when a musculoskeletal RTM device or digital therapeutic tool is supplied for a limited monitoring window within a 30-day period. Here are examples of how CPT 98985 is used in practice:
CPT 98985 Billing Requirements and Eligibility
CPT 98985 reports RTM device supply for musculoskeletal system monitoring when RTM data is captured on 2–15 days in a 30-day period. Because RTM device supply codes are threshold-based, documentation must clearly support both device supply and data availability.
Patient Eligibility Criteria
Patients appropriate for musculoskeletal RTM typically:
- Have a musculoskeletal condition or rehabilitation plan requiring monitoring
- Are enrolled in an RTM program under provider oversight
- Use a qualified RTM device or digital therapeutic tool
- Receive monitoring in a home or community-based setting
Musculoskeletal RTM may be used for:
Device and Data Requirements
To qualify for 98985:
- The RTM device/tool must support musculoskeletal therapy monitoring
- Data capture should be automated or system-generated (not manual logging)
- The device/tool must be furnished as part of the provider’s RTM program
- RTM data must be captured on 2–15 days in the billing cycle
- Data must be accessible via the RTM platform if review is required
Who Can Bill CPT 98985
CPT 98985 may be billed by:
CPT 98985 Billing Documentation Checklist
- Provider order / RTM monitoring plan
- Clinical rationale for musculoskeletal RTM
- Description of therapy or intervention being monitored
- RTM device or digital therapeutic description
- Supports MSK therapy adherence or response
- Automated data capture and transmission
- Device supply and activation record
- Date furnished to the patient
- Confirmation system access was enabled
- Transmission / data availability documentation
- Evidence of RTM data on 2–15 days
- Platform logs or usage summaries tied to the billing month
- Supervising provider identification
- Claim billed under a physician or QHP
- Attestation that:
- Only one MSK RTM device supply code was billed per 30-day cycle
- Short-duration and full-duration MSK RTM codes were not billed together
Common CPT 98985 Billing Mistakes (and How to Avoid Them)
❌ Billing Without Meeting the 2-Day Minimum
RTM data must be captured on at least 2 days in the billing period to qualify for CPT 98985.
❌ Billing Both Short- and Full-Duration MSK Device Codes
Choose the correct device supply code based on whether the patient met the 16-day threshold.
❌ Using Manually Logged Exercise Data
Manually entered patient data should not be counted toward RTM day thresholds unless supported by the RTM system.
❌ Missing Device Supply Documentation
Claims may be denied if records do not clearly show the RTM device/tool was furnished and activated.
❌ Duplicate Billing in the Same 30-Day Period
Only one musculoskeletal RTM device supply code may be billed per patient per cycle.
❌ Ignoring Therapy-Related Billing Rules
Some payers apply therapy-specific rules depending on who furnishes the RTM service; documentation should support staff roles and supervision.



