CPT 99445 Description, Billing Rules, & Use Cases
CPT 99445 is used to report the monthly supply, transmission, and monitoring of Remote Physiological Monitoring (RPM) devices when patient-generated data is collected and transmitted on 2 to 15 days within a 30-day period. This code supports RPM programs that rely on patient data collected outside the clinic—such as blood pressure, glucose, weight, or pulse oximetry—and allows providers to receive reimbursement for connected care infrastructure when the standard 16-day data threshold is not met.
CPT 99445 is billed instead of CPT 99454 for the same billing period and may not be reported concurrently with CPT 99454. It is intended for shorter-duration RPM participation while maintaining continuity of device supply and data transmission.
What is CPT Code 99445?
CPT 99445 covers the ongoing supply and management of a physiologic monitoring device used in a Remote Physiologic Monitoring (RPM) program when patient-generated data is captured and transmitted on 2 to 15 days within a 30-day period. It is billed once per 30-day period, as long as:
- A qualified RPM device is supplied to the patient
- The device is capable of automatic data transmission
- The system captures and transmits patient data on 2–15 days during the billing month
CPT 99445 does not include device setup or patient education (reported under CPT 99453), nor does it include RPM treatment management time (reported under CPT 99457, 99458, or 99470). Instead, CPT 99445 represents the passive data infrastructure that supports RPM—such as device connectivity, data transmission, portal access, and monitoring alerts—when the standard 16-day threshold required for CPT 99454 is not met.
This code is reported in place of CPT 99454 for the same billing period and may not be billed concurrently with CPT 99454.
It applies to both chronic and acute physiologic monitoring use cases, including:
- Hypertension
- Heart failure
- Diabetes
- Obesity
- COPD and post-discharge recovery
CPT 99445 Time Thresholds and Code Combinations
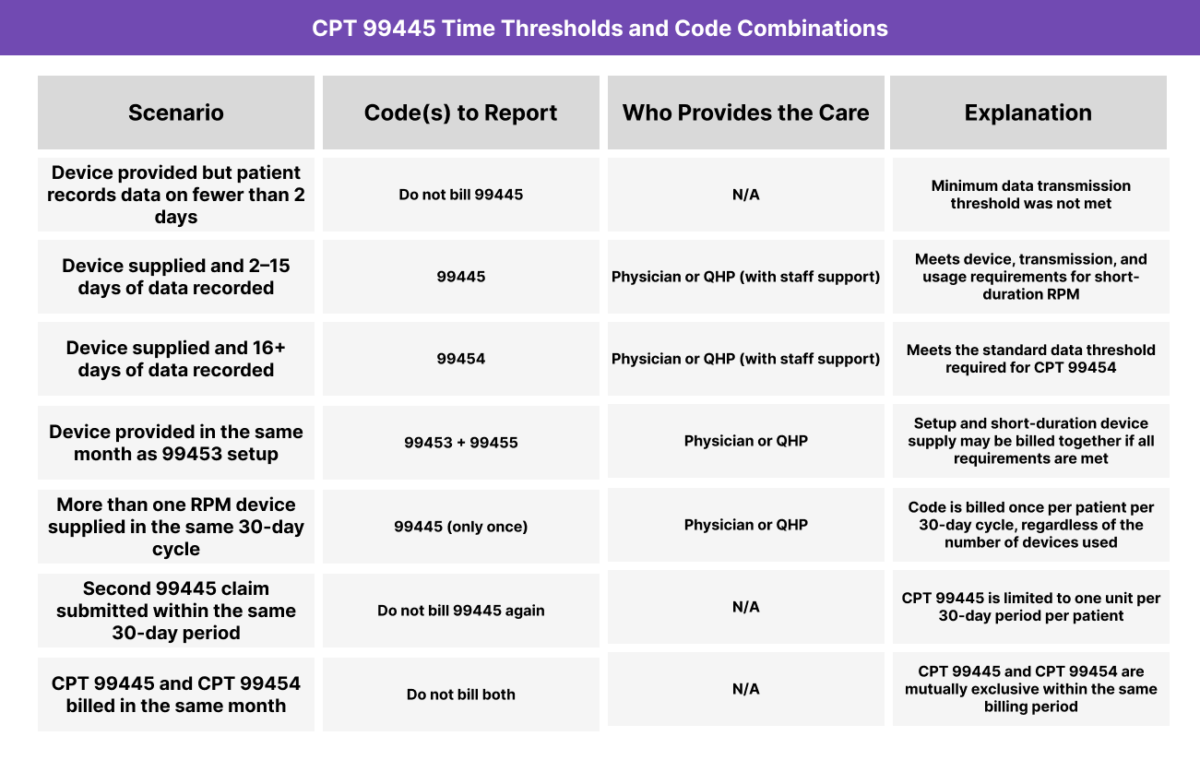
CPT 99445 may be billed once per calendar month to report the supply and transmission of a physiologic monitoring device when the minimum data threshold required for CPT 99454 is not met. Providers must confirm both technical compliance and patient engagement before submitting the claim.
Important to Note:
CPT 99445 requires:
- An FDA-defined RPM device assigned to the patient
- Device use that results in 2–15 days of data transmission within the billing month
- A single billing instance per 30-day period, per patient
- Supervised RPM infrastructure with accessible data review capabilities
When to Use CPT 99445: Common Scenarios and Use Cases
CPT 99445 is used to report the monthly device supply and data transmission component of a Remote Physiologic Monitoring (RPM) program when the patient’s device data is transmitted on 2 to 15 days during a 30-day period. It is billed once per 30-day cycle when the device is supplied and transmission occurs as expected, but the 16-day minimum required for CPT 99454 is not reached.
Here are examples of how CPT 99445 is used in practice:
CPT 99445: Billing Requirements and Eligibility
CPT 99445 is used to report the monthly supply and transmission of a Remote Physiologic Monitoring (RPM) device when patient-generated physiologic data is captured and transmitted on 2 to 15 days within a 30-day period. This code is data-threshold dependent, requires documented device supply and transmission capability, and should be tied to a physician-ordered monitoring plan.
It does not include setup or patient onboarding (CPT 99453) or provider-led RPM treatment management (CPT 99457/99458/99470). Instead, it captures the passive, ongoing service layer that allows physiologic data to flow from the patient to the care team when the standard 16-day transmission threshold for CPT 99454 is not met.
Important: CPT 99445 is billed instead of CPT 99454 for the same billing period and may not be reported concurrently with CPT 99454.
Patient Eligibility Criteria
To be eligible for RPM under CPT 99445, the patient must:
- Have a condition that warrants daily or near-daily physiologic monitoring
- Reside in a home or community-based setting (not a facility or hospital)
- Be enrolled in an active RPM program with physician oversight
Be using a qualified, FDA-defined medical device capable of data transmission
RPM is commonly used for:
- Cardiovascular disease (e.g., hypertension, CHF)
- Diabetes management
- Respiratory illnesses (e.g., COPD, post-viral recovery)
- Weight and nutrition monitoring
- Post-surgical recovery or medication titration
Device and Setup Requirements
To qualify for 99445:
- The device must be FDA-defined as a medical device
It must be capable of:
- Measuring physiologic data (e.g., BP, glucose, weight, oximetry)
- Automatically recording and transmitting data (manual entry is not allowed)
- The device must be supplied by the provider
- The patient must transmit data on 2–15 days within a 30-day period
- Data must be accessible and reviewable by the clinical team if alerts occur
Each billing cycle represents one 30-day period, and multiple devices used concurrently for the same patient are not billed separately under 99445
Who Can Bill CPT 99445
CPT 99445 may be billed by:
- Physicians
- Nurse Practitioners (NPs)
- Physician Assistants (PAs)
- Other Qualified Healthcare Professionals (QHPs)
Device distribution and monitoring may be performed by clinical staff, but billing is tied to a supervising provider under general supervision.
Billing Frequency and Data Requirements
- CPT 99445 is billed once per 30-day period, per patient
- It requires 2–15 days of data transmission during that period
- Billing is only valid if the patient actively uses the device and transmission occurs
Reimbursement generally covers:
- Cellular or Bluetooth transmission
- Portal/platform access
- Data monitoring infrastructure
- The device lease or supply
CPT 99445 Billing Documentation Checklist
To support compliant billing of CPT 99445, include the following in the patient record:
- A documented provider order for RPM services:
- Includes clinical rationale for physiologic monitoring
- Specifies the type of data to be monitored (e.g., BP, weight, glucose)
Description of the RPM device:
- Meets the FDA definition of a medical device
- Capable of automated data transmission (not manual logging)
- Assigned to the patient for use in their home or community setting
Device supply and activation record:
- Date the device was provided to the patient
- Documentation that the patient was onboarded and device functionality confirmed
- Evidence that the system was connected to a clinical monitoring platform
Transmission tracking for the billing cycle:
- Confirmation that the patient transmitted data on 2–15 days in a 30-day period
- Timeframe should match the billing period exactly
- Optional: platform-generated logs or device usage summaries
Patient participation verification:
- Confirmation that the patient engaged with the device (passively or actively)
- No extended gaps in measurement beyond allowable parameters
Supervising provider identification:
- The claim must be billed under a physician or QHP
- Clinical staff involvement must occur under general supervision
Attestation that:
- CPT 99445 was not duplicated by another provider for the same patient during the billing period
- Only one billing instance is submitted per 30-day cycle
- CPT 99445 was billed instead of CPT 99454 for the same cycle
- Devices used concurrently for the same physiologic function are not billed separately
Common CPT 99445 Billing Mistakes (and How to Avoid Them)
❌ Fewer Than 2 Days of Data Transmission
The most common mistake is billing CPT 99445 when the patient transmits data on fewer than 2 days in the billing period. Even if the device was supplied and setup occurred, CPT 99445 may not be reported unless the minimum data threshold is met.
❌ Billing CPT 99445 and CPT 99454 in the Same Month
CPT 99445 and CPT 99454 are mutually exclusive. Billing both codes for the same patient within the same 30-day period will result in denial. CPT 99445 should only be used when the 16-day requirement for CPT 99454 is not met.
❌ Manually Entered Data from Non-Qualified Devices
CPT 99445 requires automated data transmission from an FDA-defined medical device. Readings manually entered by the patient or caregiver do not qualify and should not be counted toward the data-day threshold.
❌ Duplicate Billing Within the Same 30-Day Period
Only one unit of CPT 99445 may be billed per patient per 30-day cycle. Submitting multiple claims for different devices or attempting to split billing across providers is not permitted.
❌ Device Not Supplied by the Billing Provider
The RPM device must be furnished by the billing provider or their care team. Claims may be denied if device ownership, assignment, or supply cannot be clearly documented.
❌ Using CPT 99445 When 16+ Days of Data Were Recorded
If the patient transmits data on 16 or more days in the billing period, CPT 99454 — not CPT 99445 — should be billed. Using CPT 99445 in this scenario may be considered incorrect coding.



