What Are State Plan Amendments (SPAs)?
State Plan Amendments (SPAs) are formal submissions from state Medicaid agencies to the Centers for Medicare & Medicaid Services (CMS) requesting approval to modify their Medicaid State Plan.
The State Plan functions as a comprehensive contract between each state and the federal government, outlining how the state administers its Medicaid program, including eligibility standards, payment methodologies, and benefit coverage.
When a state wishes to make a change—such as updating provider payment methods, implementing new supplemental payment programs (like DSH, UPL, or directed payments), or revising financing mechanisms (such as IGTs or CPEs)—it must submit an SPA for CMS review and approval.
Each SPA ensures that the proposed change complies with federal Medicaid law, maintains budget neutrality, and aligns with CMS oversight requirements under Title XIX of the Social Security Act.
States cannot implement most program or financing modifications until CMS formally approves the SPA, which then becomes part of the state’s permanent Medicaid plan.
SPAs are therefore the core administrative vehicle for Medicaid program updates, ensuring that all operational, fiscal, and policy changes are transparent, legally enforceable, and federally approved.
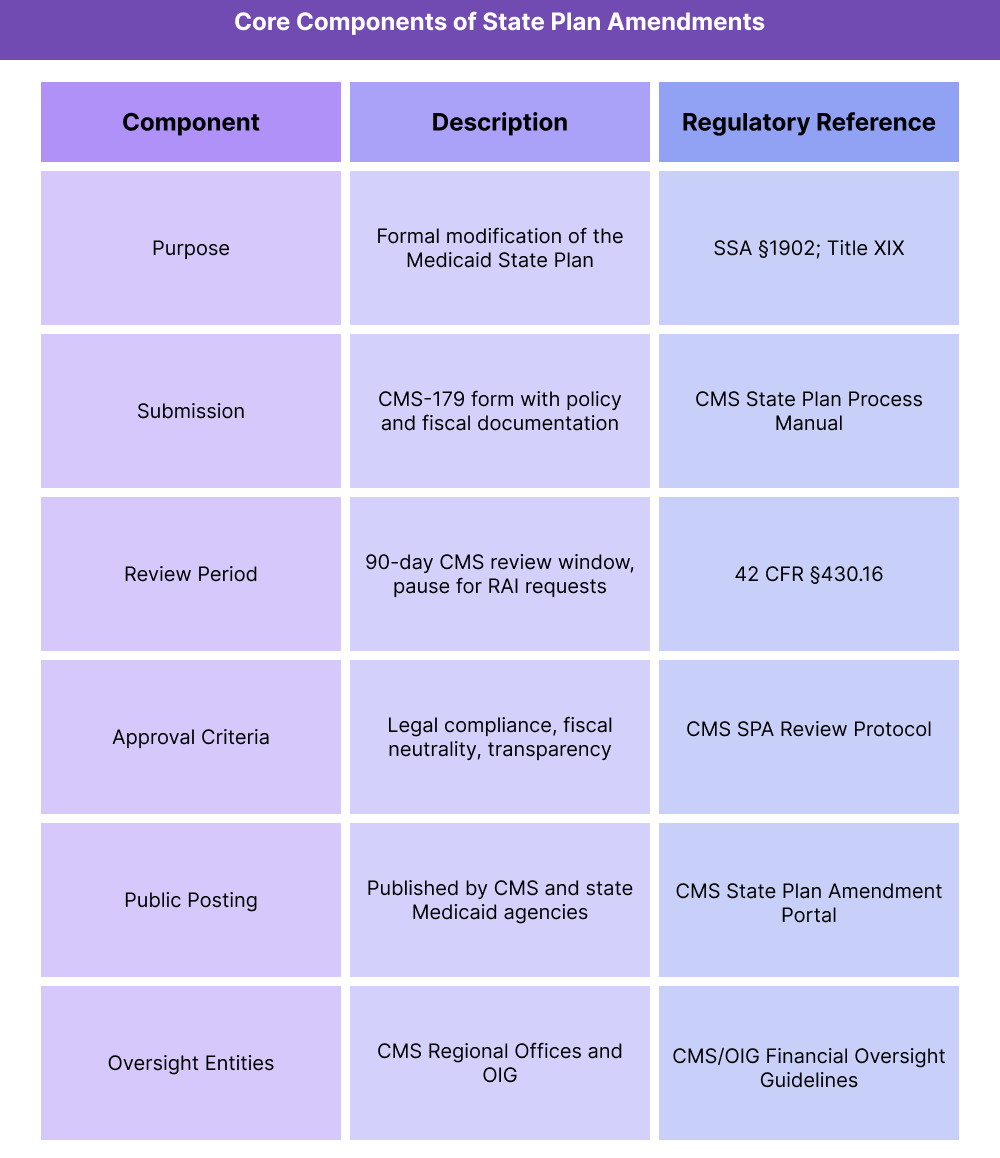
Key Components of State Plan Amendments (SPAs
State Plan Amendments (SPAs) are the formal mechanism through which states modify their Medicaid programs under federal law. Each SPA submission documents a proposed change to the State Medicaid Plan, providing CMS with the information needed to assess policy compliance, fiscal impact, and administrative feasibility.
SPAs allow states to adapt to evolving healthcare priorities, funding needs, and program reforms while maintaining transparency, accountability, and federal oversight.
1. Purpose and Policy Framework
- The Medicaid State Plan outlines how a state administers its Medicaid program in accordance with Title XIX of the Social Security Act.
- When states seek to modify coverage, provider payments, eligibility, or financing structures, they must submit an SPA to CMS for review.
- Common SPA purposes include:
- Adding or updating payment methodologies (e.g., DSH, UPL, directed payments)
- Adjusting eligibility criteria or benefit definitions
- Revising financing mechanisms, such as IGTs or CPEs
- CMS approval ensures the state remains compliant with federal Medicaid law, budget neutrality, and program integrity standards.
2. SPA Submission and Review Process
- The state Medicaid agency prepares the SPA using the CMS-179 submission form and supporting documentation.
- Each submission must include:
- A description of the proposed change
- Statutory or regulatory authority supporting the amendment
- Fiscal impact analysis and budget projections
- Implementation timeline and affected provider classes
- Once submitted, CMS has 90 days to review the SPA and either:
- Approve it,
- Request additional information (which pauses the review clock), or
- Deny it if noncompliant with federal law.
- Upon approval, the amendment becomes an official part of the state’s Medicaid plan and is enforceable under federal and state law.
3. Types of SPAs
Payment SPAs – Modify provider reimbursement methodologies, including UPL, DSH, and directed payment programs.
- Eligibility SPAs – Change population criteria, benefit coverage, or enrollment processes.
- Financing SPAs – Define or revise the use of IGTs, CPEs, or other mechanisms for state share funding.
- Administrative SPAs – Update procedural or reporting requirements for Medicaid operations.
- Emergency or Expedited SPAs – Allow time-sensitive implementation in response to federal policy shifts or public health emergencies.
4. CMS Oversight and Approval Criteria
- CMS evaluates SPAs for:
- Legal compliance – Alignment with federal statutes and regulations.
- Fiscal soundness – Consistency with federal matching rules (FMAP) and budget neutrality.
- Program integrity – Prevention of improper payments or circular funding.
- Transparency – Disclosure of affected provider classes, payment sources, and methodologies.
- CMS may issue formal Requests for Additional Information (RAIs) if clarification or documentation is needed before approval.
- Approved SPAs are published on both the state Medicaid agency’s website and the CMS Medicaid portal for public access.
5. Reporting and Maintenance Requirements
- States must maintain SPA-related documentation for at least three years after implementation.
- CMS may periodically audit SPA compliance as part of its financial management reviews or OIG evaluations.
- When states modify an existing SPA, they must submit an amendment to the amendment, not a new plan.
- States must also ensure their Medicaid Management Information System (MMIS) and CMS-64 reporting reflect approved SPA changes accurately.
How State Plan Amendments (SPAs) Work in Practice
State Plan Amendments (SPAs) are the primary mechanism for implementing Medicaid program and financing changes at the state level.
They translate state policy priorities—such as expanding eligibility, revising payment methods, or modernizing delivery systems—into federally approved operational frameworks.
Each SPA reflects a collaboration between the state Medicaid agency and CMS, ensuring that local innovations remain compliant with federal standards.
Step 1: Identifying the Policy or Financing Need
- The SPA process begins when a state identifies a policy, payment, or programmatic change requiring CMS approval.
- Common triggers include:
- Adjusting provider reimbursement (e.g., new UPL or DSH methodologies).
- Introducing a new supplemental payment or value-based program.
- Modifying eligibility criteria or benefit coverage.
- Aligning financing mechanisms (such as IGTs or CPEs) with CMS guidance.
- The state Medicaid agency develops a preliminary proposal in coordination with state budget offices, public hospitals, and other stakeholders.
Step 2: Drafting the SPA Submission
- The Medicaid agency prepares the SPA using the CMS-179 form and supporting documentation, which typically includes:
- Legal citations under the Social Security Act and state law.
- Policy and program descriptions outlining the amendment.
- Fiscal impact statements estimating federal and state share changes.
- A narrative justification of how the change complies with federal rules.
- Draft SPAs often undergo informal consultation with CMS Regional Offices before formal submission, especially for complex financing arrangements.
Step 3: CMS Submission and Initial Review
- The state submits the SPA electronically through the CMS State Plan Amendment (Medicaid SPA) Portal.
- CMS has 90 days to review the submission and may issue a Request for Additional Information (RAI) if clarifications are needed.
- During the review:
- CMS assesses the amendment for statutory compliance, fiscal soundness, and equity implications.
- States respond to RAIs to resume the review timeline.
- Once approved, CMS issues a formal approval letter and assigns the amendment an identifying SPA number (e.g., SPA 25-003).
Step 4: Implementation and Operationalization
- After CMS approval, the amendment becomes part of the official State Medicaid Plan.
- The state must update internal systems, including:
- MMIS (Medicaid Management Information System) for claims and payment adjustments.
- CMS-64 financial reporting templates for expenditure tracking.
- Provider manuals and policy bulletins reflecting new reimbursement rules.
- States also provide public notice of SPA changes, ensuring transparency for providers and stakeholders.
Step 5: Fiscal and Compliance Reporting
- Approved SPAs directly affect Medicaid financial reporting, requiring updates to:
- CMS-64 reports to reflect new spending categories or payment formulas.
- State accounting systems for monitoring federal and non-federal share contributions.
- CMS may request post-implementation reviews to confirm compliance with approved methodologies.
- States must retain SPA-related financial and policy documentation for at least three years and be prepared for OIG or CMS audits.
Step 6: Coordination with Other Medicaid Processes
- SPA development and implementation often intersect with other Medicaid approval mechanisms:
- 1115 Demonstration Waivers – for broader program reforms outside standard Medicaid rules.
- 1915(b)/(c) Waivers – for care coordination or long-term services programs.
- State budget amendments and legislative authorizations that fund SPA-driven changes.
- Coordination between these processes ensures budget neutrality, program alignment, and federal consistency.
Step 7: Ongoing Maintenance and Amendments
- States may submit subsequent amendments to modify previously approved SPAs as policies evolve.
- CMS encourages proactive communication and pre-submission discussions to expedite review.
- Annual CMS-State Plan reviews verify that all approved SPAs are current, implemented correctly, and published publicly.
SPAs in Medicaid Billing, Reimbursement, and Fiscal Limitations
State Plan Amendments (SPAs) are the regulatory gateway for Medicaid reimbursement and funding changes. They determine how states structure payments, define allowable costs, and ensure compliance with federal matching rules.
While SPAs provide flexibility for states to tailor Medicaid programs, they also introduce significant fiscal management and audit accountability requirements tied to every approved payment methodology.
How SPAs Affect Medicaid Reimbursement
- Each SPA that alters provider reimbursement—such as UPL, DSH, or value-based payment programs—directly impacts the state’s Medicaid financial flow.
- Approved SPAs establish the methodology for calculating payments, defining rate structures, or authorizing supplemental payments.
- CMS requires that SPA-based payments:
- Are economically reasonable and consistent with efficiency and quality of care.
- Adhere to budget neutrality and aggregate payment limits under federal law.
- Maintain clear documentation of both state share (e.g., IGTs or CPEs) and federal share (FMAP) funding sources.
- Once implemented, SPA methodologies become binding payment rules until modified by a subsequent amendment.
Funding Flow and Financial Reporting
- SPAs modify the state’s Medicaid expenditure structure, requiring alignment between:
- State accounting systems (budget appropriation and fund source tracking).
- CMS-64 financial reports, where expenditures must correspond to SPA-approved categories.
- Provider payment systems (MMIS) for claims, rate calculations, and supplemental payment issuance.
- Any discrepancy between approved SPA language and actual payment execution can trigger a CMS financial management review (FMR) or disallowance.
- SPAs that include complex financing mechanisms (e.g., IGTs, CPEs, or directed payments) must disclose those structures in detail to ensure federal transparency.
CMS Oversight and Fiscal Accountability
- CMS monitors SPA implementation through regional office reviews, financial audits, and routine expenditure reconciliations.
- States must provide clear evidence that payments:
- Comply with federal upper payment limits and DSH caps.
- Reflect accurate non-federal share contributions.
- Do not include impermissible provider donations or recycled funds.
- CMS and the Office of Inspector General (OIG) can request supporting documentation—including fund source certifications, payment models, and cost reports—at any time.
- In cases of noncompliance, CMS may issue a disallowance, requiring states to repay the federal share associated with the SPA.
Audit Exposure and Risk Mitigation
- SPAs involving supplemental or value-based payments carry higher audit exposure due to their fiscal complexity.
- Common audit risks include:
- Inconsistent reporting between SPA-approved and implemented payment models.
- Incomplete disclosure of IGT or CPE financing arrangements.
- Misalignment between provider payments and CMS-64 reporting.
- To mitigate these risks, states are encouraged to:
- Conduct internal financial reconciliations each quarter.
- Maintain comprehensive SPA documentation (submission, approval, implementation, and audit trail).
- Engage CMS regional offices early when pursuing complex financial or structural changes.
Fiscal Limitations and Timing Constraints
- SPAs cannot be applied retroactively unless expressly permitted by CMS, limiting reimbursement flexibility for past expenditures.
- CMS enforces strict 90-day review timelines but may pause the clock with RAIs (Requests for Additional Information), extending implementation delays.
- Unapproved or pending SPAs prevent states from claiming federal matching funds (FMAP) for the proposed expenditures.
- Budget forecasting and cash flow planning must account for federal review cycles to avoid reimbursement gaps.
Policy and Reform Considerations
- CMS continues to refine the SPA process to improve transparency, standardization, and equity across state Medicaid programs.
- Key trends include:
- Public posting of SPA methodologies and fiscal impact summaries.
- Increased scrutiny of financing arrangements using IGTs and CPEs.
- Integration of value-based payment models into SPA submissions to align reimbursement with quality performance.
- These reforms aim to ensure that SPAs remain both a flexible policy tool and a rigorously accountable fiscal instrument in Medicaid administration.
Key Takeaway
SPAs define the financial backbone of Medicaid reimbursement, shaping how funds flow from CMS to providers.
They provide flexibility for innovation but demand meticulous compliance, documentation, and coordination between state finance offices and federal reviewers.
Properly structured SPAs ensure fiscal integrity, program transparency, and continued access to federal funding—the cornerstones of sustainable Medicaid operations.
How SPAs Influence Quality, Access, and Equity in Medicaid Financing
State Plan Amendments (SPAs) are the regulatory foundation for shaping Medicaid program design, funding, and accountability.
Every SPA submission and approval not only adjusts state policy but also directly affects provider reimbursement fairness, beneficiary access, and health equity outcomes.
By aligning state Medicaid operations with CMS’s modernization and equity goals, SPAs serve as both a compliance tool and a catalyst for continuous quality improvement.
Promoting Access Through Program Flexibility
- SPAs enable states to respond quickly to emerging healthcare needs—such as expanding coverage, updating telehealth policies, or addressing workforce shortages—while remaining under federal oversight.
- Through the SPA process, states can direct funding to safety-net hospitals, rural facilities, and underserved regions, ensuring that care remains accessible across geographic and socioeconomic lines.
- Because SPAs are subject to CMS approval, they ensure that access-related changes align with federal beneficiary protection standards under Title XIX.
- Flexibility balanced with accountability allows states to innovate without compromising Medicaid’s foundational equity mission.
Advancing Quality Through Payment and Policy Reform
- Many SPAs now incorporate value-based payment methodologies and quality-linked reimbursement systems that reward measurable improvements in care outcomes.
- CMS encourages states to embed performance metrics, clinical benchmarks, and equity indicators into SPA-driven payment structures.
- By linking reimbursement to performance, SPAs help shift Medicaid financing from volume-based to value-based care delivery, improving clinical quality and fiscal efficiency.
- Examples include SPAs that establish directed payments tied to hospital quality metrics, population health outcomes, or readmission reduction goals.
Ensuring Equity Through Federal Oversight and Transparency
- SPAs are a key lever for advancing equitable healthcare delivery by requiring federal review of all program and financing changes.
- CMS’s SPA approval process helps prevent regional disparities in Medicaid access or payment methodologies.
- States must publicly post SPA submissions and approval letters, enabling transparency for providers, policymakers, and the public.
- This visibility helps ensure that changes to eligibility, reimbursement, or provider classes promote consistent treatment across populations and geographies.
Integrating Equity Frameworks into SPA Policy Design
- CMS increasingly encourages states to align SPAs with health equity frameworks, prioritizing interventions that reduce disparities in outcomes and access.
- Examples include:
- SPAs expanding postpartum coverage for Medicaid beneficiaries.
- SPAs funding care coordination programs for high-need, high-cost populations.
- SPAs enhancing payment for behavioral health or rural access initiatives.
- These amendments illustrate how SPA policy serves as a direct mechanism for advancing social and economic equity within the Medicaid system.
Transparency and Accountability as Quality Drivers
- Because every SPA must include fiscal impact analysis, legal authority, and implementation details, the process itself enforces operational transparency.
- Public documentation of SPA methodologies allows for external evaluation of program fairness and performance.
- This transparency aligns with CMS’s commitment to data-driven policy evaluation, where funding, quality, and equity can be monitored across states.
- States that leverage SPAs strategically can use this visibility to demonstrate compliance excellence and build stronger cases for federal innovation waivers.
The SPA’s Role in CMS Modernization and Equity Goals
- SPAs have evolved into one of CMS’s primary tools for implementing national priorities such as:
- Health equity advancement under the CMS Framework for Health Equity.
- Digital modernization and interoperability through state-level policy updates.
- Payment reform alignment with federal value-based purchasing initiatives.
- By requiring every policy change to undergo a structured, transparent approval process, SPAs ensure that equity, quality, and fiscal responsibility remain embedded in Medicaid program design.
Frequently Asked Questions about SPAs
1. What is a State Plan Amendment (SPA)?
A State Plan Amendment (SPA) is a formal request submitted by a state Medicaid agency to the Centers for Medicare & Medicaid Services (CMS) to modify its Medicaid State Plan.
SPAs are required whenever a state wants to change eligibility standards, benefits, payment methodologies, or financing mechanisms. Once approved, the SPA becomes part of the state’s permanent Medicaid framework.
2. When is an SPA required?
States must submit an SPA whenever they propose:
- A new or revised payment methodology, such as UPL, DSH, or value-based payments.
- A change in provider reimbursement or benefit coverage.
- A modification of eligibility rules, program administration, or financing structures like IGTs or CPEs.
Essentially, any material policy or fiscal change in Medicaid operations requires CMS approval through an SPA.
3. How does the SPA approval process work?
- The state prepares and submits the amendment using the CMS-179 form and supporting documentation.
- CMS has 90 days to review the submission but may pause the clock if it issues a Request for Additional Information (RAI).
- Once approved, CMS sends a formal approval letter and publishes the SPA on its public portal.
- The approved amendment immediately becomes part of the state’s official Medicaid State Plan.
4. How are SPAs different from waivers?
- SPAs modify a state’s Medicaid plan within the limits of federal law under Title XIX.
- Waivers—such as 1115 Demonstration Waivers or 1915(b)/(c) Waivers—allow states to waive certain federal requirements to test new delivery or payment models.
In short, SPAs are permanent policy updates, while waivers are time-limited demonstrations requiring periodic renewal.
5. What documentation is required in an SPA submission?
Each SPA must include:
- The CMS-179 cover form.
- A detailed description of the proposed change and its policy rationale.
- The statutory and regulatory authority supporting the amendment.
- A fiscal impact analysis projecting changes to the state and federal share of Medicaid spending.
- Implementation timelines and affected provider or beneficiary groups.
6. What happens if a state implements a change without an approved SPA?
If a state implements a Medicaid policy change before CMS approval, federal matching funds (FMAP) may be disallowed for related expenditures.
CMS can require the state to repay the federal share, issue a corrective SPA, or suspend funding for the affected program.
Pre-implementation without approval is considered a compliance violation under federal Medicaid law.
7. Why are SPAs important for Medicaid transparency and equity?
SPAs ensure that every Medicaid program change—whether financial, clinical, or operational—is federally reviewed, publicly documented, and legally enforceable.
This process upholds equity, accountability, and consistency across states, ensuring that payment and policy changes are implemented transparently and align with CMS’s national quality and health equity goals.
